Spectrometer
Public Lab's Do-It-Yourself spectrometers are designed to help everyday people detect pollutants where they live.
Our community has been working since the 2010 Deepwater Horizon oil spill to develop a cheap, open source, Do-It-Yourself spectrometer which we hope to use to identify oil pollution in soil and water, as well as a range of other possible contaminants.
What's spectrometry?
Colored light is often a blend of different colors. A spectrometer is a device which splits those colors apart, like a prism, and measures the strength of each color. A typical output of a spectrometer looks like this spectrum of the daytime sky, with the actual light spectrum at the top and the graph of wavelength (horizontal axis) and intensity (vertical axis) below:
Types of spectrometry
There are different ways to use spectrometers, and the key difference is how you illuminate your sample.
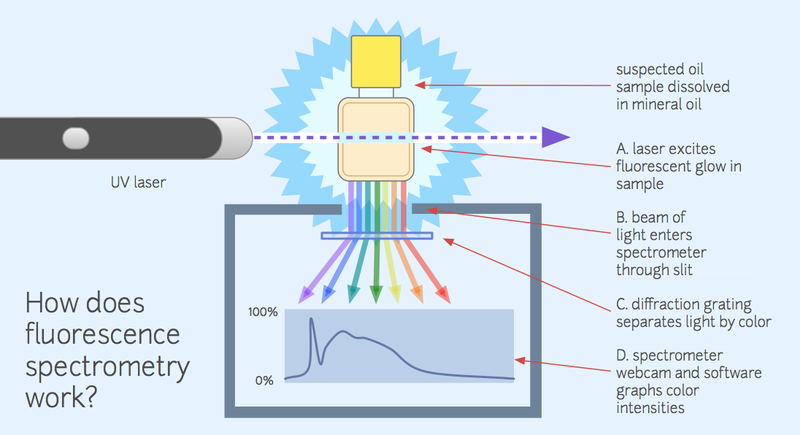
Fluorescence
This project focuses on fluorescence spectrometry in order to identify oil pollution samples, which is where a high-energy light like an ultraviolet laser is used to excite a sample so that it fluoresces, or glows.
See the lead image of this page for a diagram of a fluorescence spectrometer setup. Since different oils fluoresce in different colors, this technique can be used to match an unknown sample with a reference sample to identify it.
Read more on the Oil Testing Kit page »
Emission
Emission spectroscopy is the kind often done in the classroom, where burning a material emits a colored flame. A spectrum of this colored flame can be used to match a material, but it can be unsafe to burn unknown samples, so we have primarily begun to use this technique to attempt to monitor distant flares, for example at gas refineries in Louisiana, to try to detect heavy metals.
[image of refinery watching]
Absorption
Absorption spectroscopy -- shining a full-spectrum light like a halogen or incandescent (not a fluorescent or laser) through a sample to see what colors are absorbed -- is a bit more difficult in the visible light range, as most of the "fingerprint" features of spectra are too long or too short wavelengths for our webcam-based devices. However, a considerable amount of work has been done on absorption spectrometry of:
Make a spectrometer
The links at the top of the page offer step-by-step instructions on making your own spectrometer. Our main design, the Desktop Spectrometer features:
- around 400-900 nanometer range, maybe wider (what you can see with the naked eye, plus some infrared)
- 1-5 nm spectral resolution
- 20-30 samples per second
- ~ $15 in materials
- < 1 hour construction time
- web-based, open-source software
Upgrades
Once you've built a spectrometer, there are many ways to improve it -- by using a narrower slit, darkening the interior, using a better camera, and more.
For upgrading the USB webcam-based Desktop Spectrometry Kit, see http://publiclab.org/wiki/dsk#Upgrades
Software
Along with the physical devices, the Public Lab community has also developed Spectral Workbench, an website to capture data with your spectrometer, analyze and compare spectra, share them in an open database, and comment and collaborate with others.
The software includes:
- direct connection to your USB-based or smartphone-based device
- calibration, comparison, and matching tools
- XML, JSON, and CSV data download
- a JavaScript API
- offline mode
- read more in the documentation
FAQ
Frequently Asked Questions can be found here »
Challenges
This document, and this methodology, is still under active development. What you see on this page is only the best attempt so far at collating and presenting the work of Public Lab contributors to date. Some of the challenges that remain include:
- easy auto-calibration of the device (see progress by @sreyanth)
- easier and more consistent sample preparation methods
- additional challenges specific to oil identification can be found on the Oil Testing Kit page
Other uses
While many of us have focused on identifying oil pollution with fluorescence spectrometry, there are many other uses for cheap, open source spectrometers, and many other ways to use a spectrometer.
Kickstarter
In 2012, Public Lab ran a Kickstarter project to distribute an early version of our DIY spectrometers to over 1600 people. The video is a bit out of date, but is still a compelling way to understand what we're attempting to do:
The Homebrew Sensing Project is made possible in part by the generous support of the John S. and James L. Knight Foundation, Knight News Challenge: Health.






